|
12/28/2023 0 Comments So2 molecular geometry shape  
SO2 gives a weak acid solution when dissolved in water. It is a colorless, toxic, and inorganic gas with a pungent smell like Nitric acid. SO2 (Sulfur dioxide) is the entity of a bond between Sulfur and Oxygen atoms. They are found in the outermost electron shell of atoms and can be identified by drawing lewis’s structure. They are also called lone pairs of electrons. Unbonded pairs of electrons are unshared valence electrons. The valence electrons of oxygen in SO2 are 6.The sulfur valence electron is equal to 6.The three pairs of bonding electrons are arranged in the plane at an angle of 120 degrees. 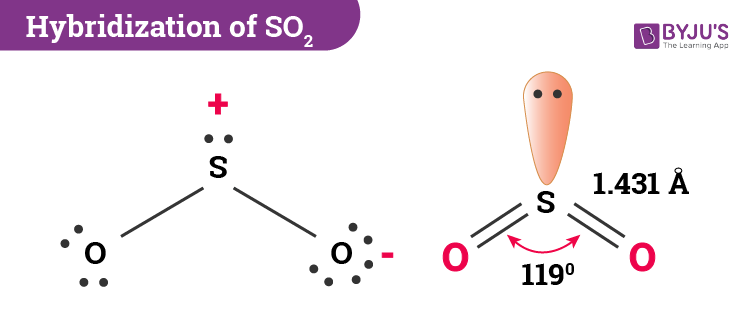
The molecular geometry of SO 2 is a trigonal planner.Related Links CO 2 Lewis Structure and Molecular Geometry SO 2 (Sulfur Dioxide) Lewis structure N 2O Lewis Structure| Laughing Gas CLF3 Lewis Structure, Molecular Geometry, and Polarity How Many Water Bottles is a Gallon| Examples HCN Lewis Structure & Molecular Geometry There are four lone electron pairs in the oxygen molecule.O2 is a nonpolar molecule with linear geometry.The bond angle is 180 degrees, and there are 12 valence electrons.In the O2 Lewis structure, two oxygen atoms are connected together with a double bond.To summarize everything in this article, the following are some important points: However, burning occurs only when the mixture of fuel and oxygen is sufficiently heated. Burning requires the presence of oxygen.It is paramagnetic in nature and has two oxidation states.It is sparingly soluble in water and does not react with water.Silica, which is found in flint quartz, contains more than 50% oxygen.Calcium carbonate, which occurs in chalk and limestone marble, contains 48% oxygen.Water contains nearly 89% of combined oxygen.It is a gas at room temperature, and about 1/4 of the atmosphere by weight consists of oxygen.Respiration, the process of transferring energy from glucose to cells, requires oxygen.Oxygen comprises about 50% of the earth’s crust.It has two oxygen atoms in each of its molecules. At room temperature, oxygen is a non-metal element that exists as a gas.Oxygen has two allotropic forms, O2 and O3.O2 is a colorless, odorless, and tasteless gas.In the O2 Lewis structure, there is a double bond between two oxygen atoms.In the O2 molecule, both oxygen atoms have equal electronegativity and both atoms share equal ratios of bonded shared electrons, and the overall molecule turns out to be nonpolar in nature. Oxygen is a diatomic molecule with linear molecular geometry and bond angles of 180 degrees. These four valence electrons form two shared pairs of covalent bonds, providing a stable structure for the oxygen molecule. Step-3: Place remaining electrons around the other atomsĪs the oxygen atom requires only two valence electrons, it readily shares electrons with the neighboring oxygen atom to complete its octet. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
